NOTE: This article is the fifth in a series of 10 articles and is part of our Economic Evaluation in Healthcare 101 course. You can find a course overview and links to all 10 course modules here:
- Economic Evaluation in Healthcare 101: Course Overview
- Introduction to Economic Evaluation in Healthcare
- Types of Economic Evaluation in Healthcare
- Measuring Costs in Healthcare
- Measuring Health Outcomes
- Decision Analytic Modeling in Economic Evaluation
- Data Sources for Economic Evaluation in Healthcare
- Interpreting and Applying Economic Evaluation Results in Healthcare
- Economic Evaluation in Health Technology Assessment (HTA)
- Ethical and Equity Considerations in Economic Evaluation
- Future Trends in Economic Evaluation in Healthcare
Decision Analytic Modeling in Economic Evaluation
Decision analytic modeling is a cornerstone of economic evaluation in healthcare, enabling researchers and policymakers to simulate the clinical and economic consequences of different health interventions over time. Unlike clinical trials, which are limited in scope, duration, and generalizability, models synthesize data from diverse sources to estimate long-term costs and outcomes in a structured, reproducible way. Two commonly used modeling frameworks are decision trees and Markov models, and the application of techniques such as sensitivity analysis and discounting enhances the robustness and relevance of model-based evaluations.
1. Introduction to Decision Trees
Decision trees are graphical representations of clinical and economic pathways that patients might follow as a result of different healthcare choices. They are structured around decision nodes (choices), chance nodes (uncertain events), and terminal nodes (final outcomes).
- Decision nodes (represented by squares) – where a choice is made between alternative interventions
- Chance nodes (represented by circles) – where probabilities are assigned to different outcomes (e.g., treatment success vs. failure)
- Terminal nodes – where the final health outcomes and costs are calculated
Key features of decision trees:
- Best suited for short-term, one-time decisions with a limited time horizon.
- Useful in diagnostic test evaluations, vaccination decisions, and surgical interventions where outcomes occur quickly.
- Each branch of the tree represents a sequence of events (e.g., test → diagnosis → treatment → outcome), and expected costs and outcomes are calculated by aggregating probabilities and values along each path.
For example, a decision tree analyzing influenza vaccination may compare two options: (1) vaccinating a patient or (2) not vaccinating. Each branch would include probabilities for contracting influenza, experiencing complications, requiring hospitalization, and associated healthcare costs. The expected value (cost-effectiveness ratio) of each option is then computed by multiplying probabilities with outcomes and summing across branches.
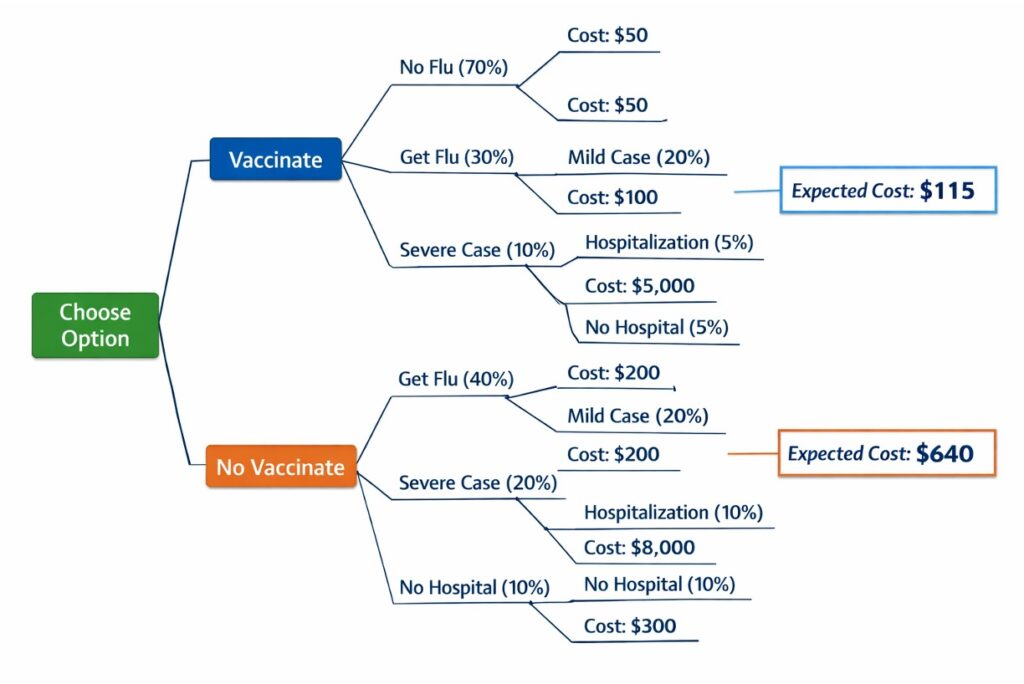
Limitations:
- As complexity and time horizons increase, decision trees become unwieldy.
- They cannot easily represent recurrent events or chronic conditions—limitations addressed by Markov models.
Reference: Hunink MGM, Weinstein MC, Wittenberg E, et al. (2014). Decision Making in Health and Medicine: Integrating Evidence and Values, 2nd ed.
2. Markov Models in Healthcare
Markov models extend decision trees by allowing patients to transition between defined health states over multiple time cycles, making them well-suited for chronic diseases and long-term interventions.
Key components:
- Health states: Mutually exclusive categories (e.g., healthy, diseased, hospitalized, dead).
- Cycle length: A fixed time interval (e.g., monthly, yearly) during which transitions between states can occur.
- Transition probabilities: Likelihoods of moving from one state to another within a cycle.
Markov models are especially valuable when:
- The disease involves recurrence or progression, such as cancer, HIV, or diabetes.
- Long-term costs and benefits need to be modeled beyond trial data.
- Outcomes accrue over time, requiring modeling of life expectancy, QALYs, and cumulative costs.
A model evaluating anticoagulation therapy in atrial fibrillation may include states like “No event,” “Stroke,” “Bleeding,” and “Death,” with transition probabilities derived from clinical studies.
A Markov model evaluating diabetes management might include health states such as “controlled diabetes,” “complications,” and “death.” Each year, a patient could remain in the same state or transition to a worse condition based on probabilities derived from clinical data. Markov models are widely used for evaluating chronic conditions like cancer, cardiovascular disease, and long-term drug treatments.
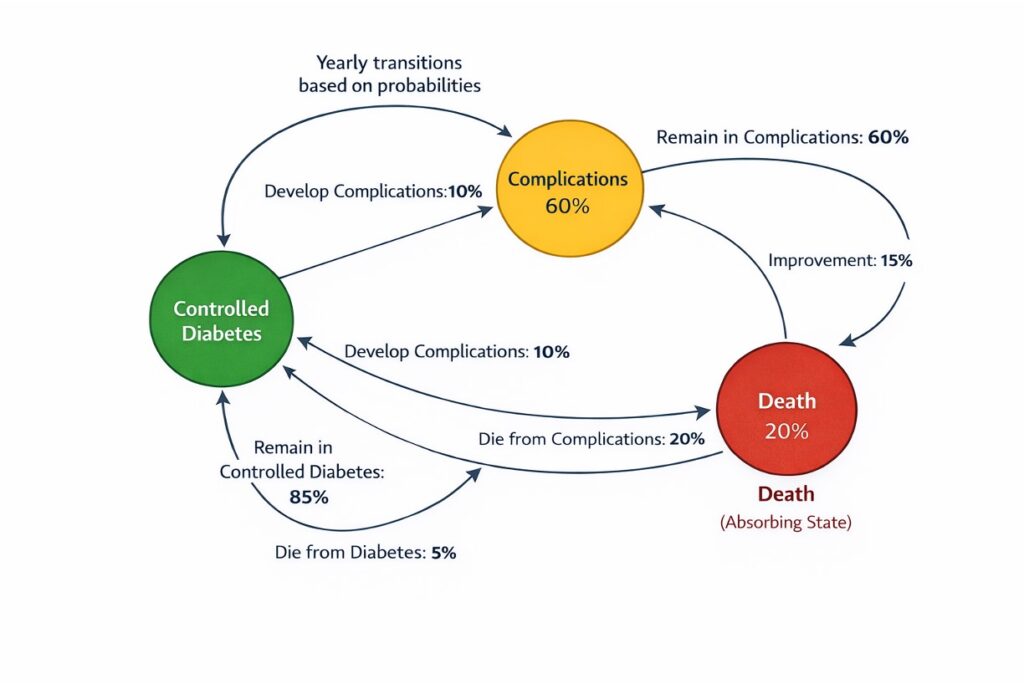
Challenges include:
- Requires significant data inputs (transition probabilities, utilities, costs).
- Risk of structural uncertainty if the model oversimplifies real-world complexity.
- A limitation of Markov models is the Markovian assumption, which states that transitions depend only on the current state and not on past history. To address this, more advanced models, such as semi-Markov or microsimulation models, can be used.
Reference: Briggs A, Claxton K, Sculpher M. (2006). Decision Modelling for Health Economic Evaluation.
3. Handling Uncertainty: Sensitivity Analysis
Uncertainty is inherent in all economic evaluations due to imperfect data, assumptions, and variability in clinical outcomes. Sensitivity analysis explores how changes in inputs affect the model’s results, helping assess robustness and decision confidence.
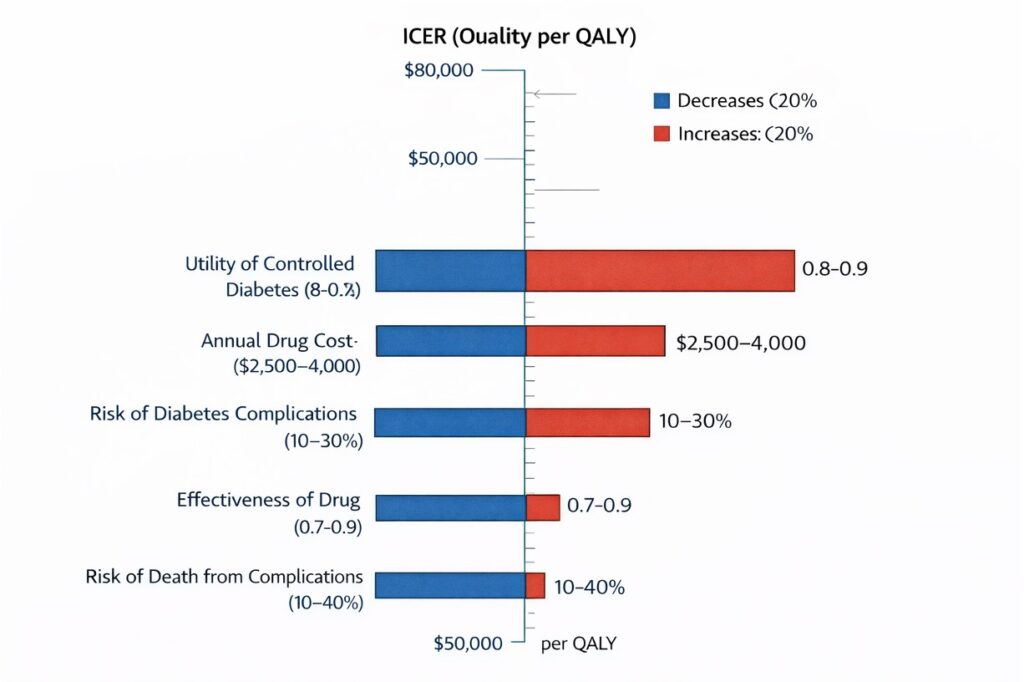
One-Way Sensitivity Analysis
- Varies one parameter at a time (e.g., drug cost, mortality rate) across plausible ranges.
- Useful for identifying key drivers of cost-effectiveness.
- Results are often presented as tornado diagrams showing the impact of each parameter.
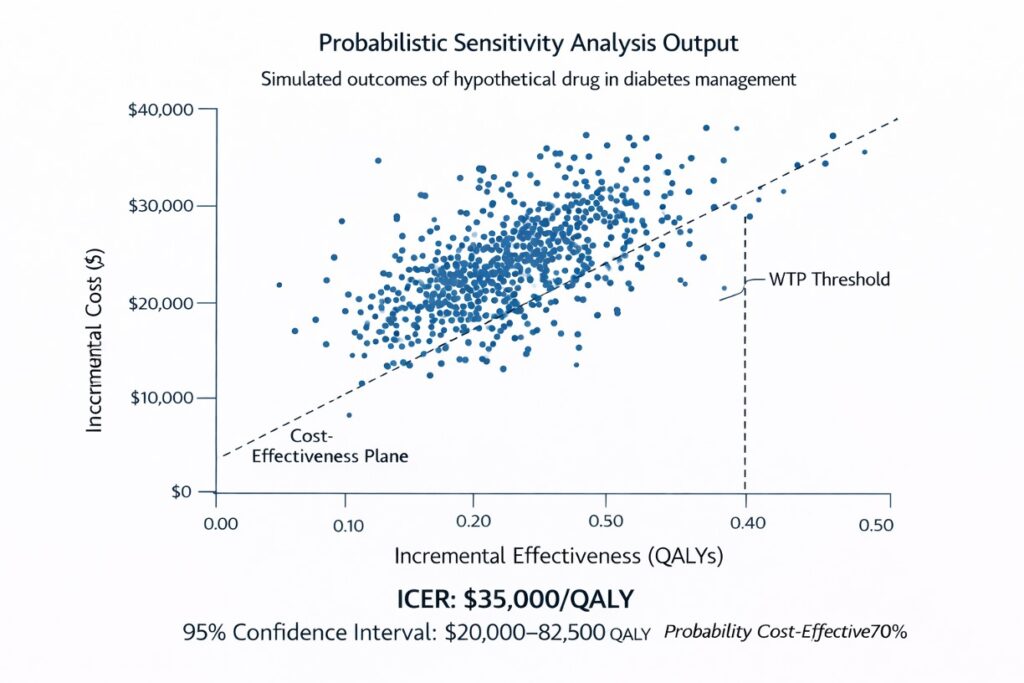
Probabilistic Sensitivity Analysis (PSA)
- Simultaneously varies multiple inputs by assigning probability distributions (e.g., beta for probabilities, gamma for costs).
- Uses Monte Carlo simulation to run thousands of model iterations.
- Produces outputs like cost-effectiveness acceptability curves (CEACs), showing the probability that an intervention is cost-effective at different willingness-to-pay thresholds.
PSA is considered the gold standard for handling parameter uncertainty in HTA contexts.
Reference: Briggs AH, Weinstein MC, Fenwick EAL, Karnon J, Sculpher MJ, Paltiel AD. (2012). Model parameter estimation and uncertainty analysis. Value in Health, 15(6), 835–842.
4. Discounting Future Costs and Benefits
In economic evaluation, health benefits and costs that occur in the future are typically discounted to reflect time preference—i.e., the principle that people prefer benefits now rather than later, and costs incurred today weigh more heavily than those in the future.
Standard practice:
- Discount rate: Commonly 3%–5% annually for both costs and outcomes, although this varies by country.
- Discounting ensures that interventions with long-term benefits (e.g., preventive care, vaccination) are not unfairly favored or penalized compared to short-term treatments.
Calculation:
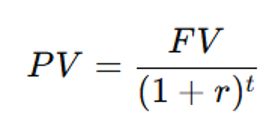
where:
- PV = Present Value
- FV = Future Value
- r = Discount rate (commonly 3-5% per year)
- t = Number of years in the future
For example, if a new cardiovascular drug prevents heart attacks 10 years from now, the health benefits must be discounted to compare them fairly with immediate costs. Similarly, future medical costs (e.g., treating complications later in life) are also discounted.
Different countries adopt different discount rates based on economic conditions. The World Health Organization (WHO) recommends a 3% discount rate, while some guidelines (e.g., NICE in the UK) use lower rates for non-monetary health outcomes. The choice of discount rate can significantly impact cost-effectiveness results, influencing whether a treatment is considered worthwhile.
Critics of discounting argue that it undervalues long-term public health interventions, such as vaccination programs or childhood nutrition initiatives, which have benefits that materialize decades later. Some propose using differential discounting, applying lower rates for health benefits compared to costs.
Policy implications:
- Different discount rates can significantly alter cost-effectiveness conclusions, especially in models with long time horizons.
- Agencies such as NICE (UK) or ICER (US) have guidelines specifying discount rates to ensure comparability across evaluations.
Reference: Drummond MF, Sculpher MJ, Claxton K, et al. (2015). Methods for the Economic Evaluation of Health Care Programmes, 4th ed.
Conclusion
Decision analytic models are essential tools in economic evaluation, allowing for the synthesis of evidence, extrapolation beyond trial data, and comparison of healthcare interventions in a structured manner. Decision trees are ideal for short-term decisions, while Markov models accommodate chronic conditions and long-term outcomes. Addressing uncertainty through sensitivity analysis and properly discounting future values are critical to producing credible and policy-relevant results. As healthcare systems increasingly rely on economic modeling to guide investment, the rigor and transparency of these models remain paramount.
References
- Briggs AH, Claxton K, Sculpher MJ. (2006). Decision Modelling for Health Economic Evaluation. Oxford University Press.
- Hunink MGM, Weinstein MC, Wittenberg E, et al. (2014). Decision Making in Health and Medicine: Integrating Evidence and Values, 2nd ed. Cambridge University Press.
- Drummond MF, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. (2015). Methods for the Economic Evaluation of Health Care Programmes, 4th ed. Oxford University Press.
- Briggs AH, Weinstein MC, Fenwick EAL, et al. (2012). Model parameter estimation and uncertainty analysis. Value in Health, 15(6), 835–842.
